Despite a few positive trends in pharmaceutical industry marketing, the total cost of prescription drugs in the U.S. rose again last year by 8.5 percent and reached a record level of $310 billion, according to a new study by IMS Health.
Spending on prescription drugs – especially the newer, breakthrough biometric drugs for the treatment of cancer, autoimmune diseases and Hepatitis-C – increased by double digits for a second year in a row and showed no sign of abating through 2020. That means that consumers, insurers and federal health care services such as Medicare, Medicaid and the Department of Veterans Affairs will be squeezed as more and more seniors and veterans seek coverage and services.
Related: Extreme Rise in Some Drug Prices Reaches a Tipping Point
“Longer-term trends driven by the Affordable Care Act and in response to rising overall healthcare costs continued to play out in 2015,” the report states. “In particular, healthcare is being delivered by different types of healthcare professionals and in different facilities, and patients face higher out-of-pocket costs as well as access barriers.”
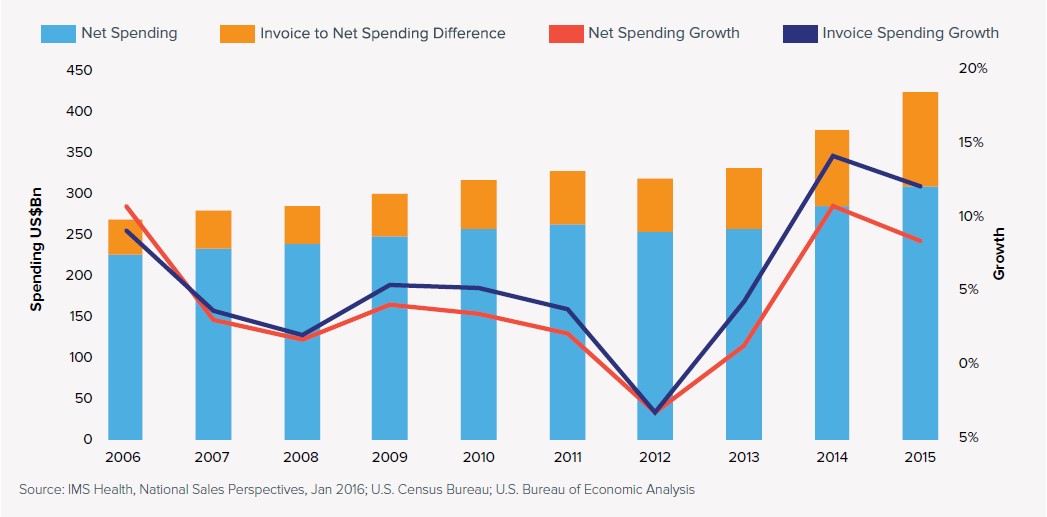
Even with its mixed forecast for industry pricing practices in the coming years, the report did provide some encouraging news. For one, the rate of growth in spending on drugs actually slowed slightly from the previous year, 2014, when prescription drug prices rose by 14.2 percent. That dip resulted from the expiration of patents that allowed cheaper generic drugs to enter the market.
Moreover, many drug manufacturers seeking to improve their tarnished reputation for price gouging, and under pressure from health care providers, continued to offer patients, insurers and health care providers substantial discounts or rebates to reduce the sting of the overall cost. When those rebates and other price concessions were factored in, net spending on drugs totaled just $310 billion last year, or an 8.5 percent increase over the 2014 level.
While the average “list price” for brands still protected by patents rose by 12.4 percent last year, the increase in the net price after discounts and other breaks was just 2.8 percent.
Related: Drug Company Profits Soar as Taxpayers Foot the Bill
“The surge of new medicines remained strong and the use of recently launched brands remained at historically high levels, while the savings from brands facing generic competition were relatively low,” according to the study, which was released on Wednesday. “Specialty drug spending on a net price basis reached $121 billion, up more than 15 percent from 2014.”
The cost of breakthrough specialty drugs doubled over the past five years; they make up a huge share of the overall annual cost of drugs. For example, $39.1 billion was spent last year on cancer drugs alone, followed by $30.2 billion for autoimmune disorders and $18.8 billion for the treatment of hepatitis.
Yet the overall cost of some of the newer breakthrough drugs, such as Gilead Science Inc.’s Sovaldi and Harvoni for the treatment of the Hepatitis-C virus, appears to have leveled off, as demand for the drug begins to subside.
Harvoni was Gilead Sciences’ top-selling drug in the U.S. last year, raking in an estimated $14.3 billion in sales before discounts, according to The Wall Street Journal. The average 12-week treatment of Sovaldi or Harvoni can cost as much as $60,000 to $94,000 before the discounts.
Related: Wonder Drugs Blow a $1 Billion Hole in VA’s Budget
While 250,000 patients were treated with Harvoni in 2015 for the deadly liver disease, up from 170,000 the year before, “the number of people beginning the therapy slowed as the year progressed, suggesting many of those most in need of the medicines may have received them,” according to the report.
Abb-Vie Inc.’s Humira and Amgen Inc.’s Enbrel – used in the treatment of inflammatory conditions including arthritis – were the second and third highest selling drugs in the country, according to The Wall Street Journal.
Since 2005, spending on prescription drugs has steadily risen with just one exception, in 2013, when it actually dipped by 3.2 percent. The new report provides only faint hope of a moderation in pricing over the next several years, projecting a mid-single digit growth rate. New innovative and very costly drug treatments will win approval from the Food and Drug Administration and continue to put pressure on overall drug spending while the introduction of more generic drugs will soften the overall impact.
Related: The Surprisingly High Cost of Beating Cancer
The drug industry has come under intense fire – and federal and congressional overview and probes – because of soaring prices of prescription drugs. The pricing practices of Gilead, Turing Pharmaceuticals, Valeant Pharmaceuticals and others -- have prompted strong criticism from the two Democratic presidential candidates, Hillary Rodham Clinton and Bernie Sanders, as well as from consumer advocates and other public policy makers who are demanding cost constraints.
The Pharmaceutical Research and Manufacturers of America and other industry advocates have justified the price structure as essential to defraying the costs of research and development, although critics often scoff at this defense and question how much of the profits are plowed back into research.
PhRMA, the industry’s chief lobbying arm, issued a statement yesterday saying that the new IMS report confirms that drug costs have begun to moderate “due to a competitive marketplace for medicines where large, powerful purchasers negotiate aggressively and generic utilization rates are nearly 90 percent.”





